Spravato® (Esketamine)
Nasal Spray
Spravato is an FDA-approved esketamine nasal spray for adults with treatment-resistant depression and major depressive disorder with suicidal thoughts. Administered in-office under medical supervision. Covered by most major insurance — including Medicaid and Medicare.
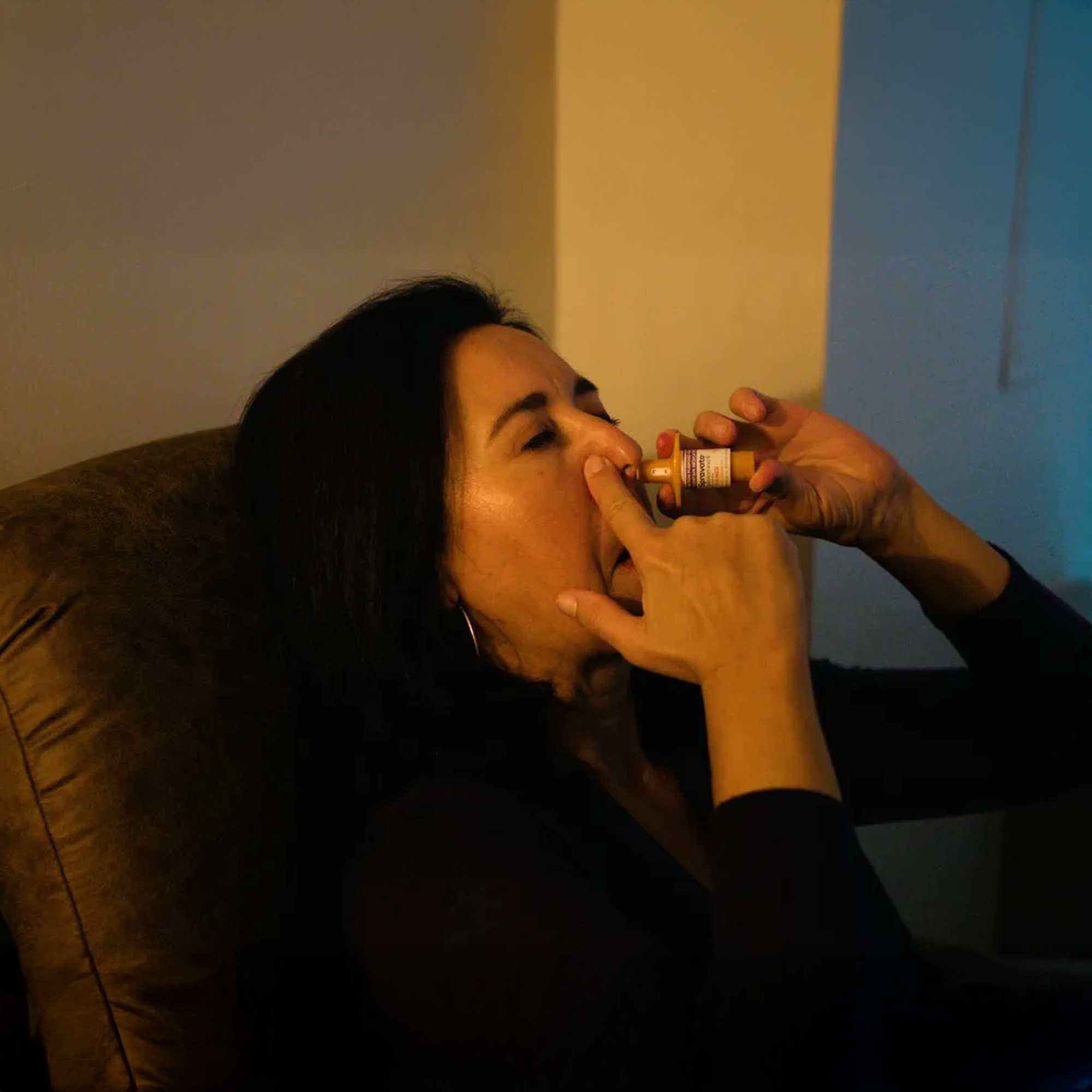
FDA-Approved Nasal Spray
Not an IV. Not an injection. FDA-approved.
Fast-Acting Relief
Some patients feel a shift the same day
Covered by Insurance
Including Medicaid & Medicare
Understanding Spravato
What Is Spravato (Esketamine)?
Spravato is the brand name for esketamine — an FDA-approved nasal spray used to treat treatment-resistant depression (TRD) and major depressive disorder (MDD) with active suicidal ideation.
Unlike traditional antidepressants, which take 4–6 weeks to work and target serotonin, Spravato works on the glutamate system — the brain’s most abundant neurotransmitter. It’s thought to help the brain rapidly form new neural connections, which is why some patients notice improvement within hours of their first dose.
It’s a nasal spray, not an IV drip or injection. It’s administered by our clinical staff at our Rockville office under medical supervision. After each dose, you stay for a 2-hour monitoring period. Then someone drives you home.
Conditions Treated
What Can Spravato (Esketamine) Treat?
Spravato has two FDA-approved uses, and growing clinical evidence for additional conditions.
Treatment-Resistant Depression (TRD)
This is Spravato’s primary FDA-approved indication. If you’ve tried two or more antidepressants and they haven’t worked — or stopped working — you likely meet the criteria. Spravato is taken alongside an oral antidepressant, not instead of one.
Major Depression with Suicidal Ideation
Spravato is one of very few treatments FDA-approved specifically for adults with MDD who are actively experiencing suicidal thoughts. Its rapid onset — often within hours — is what makes it uniquely valuable for this indication, where waiting weeks for an antidepressant to work isn’t an option.
Spravato for Anxiety
Spravato isn’t FDA-approved specifically for anxiety, but many patients with anxious depression — depression with significant anxiety symptoms — see improvement in both. The glutamate pathway it targets plays a role in anxiety regulation as well. This is something we evaluate on a case-by-case basis.
Spravato for PTSD
Research into esketamine for PTSD is still developing, but early results are promising — particularly for patients whose PTSD overlaps with treatment-resistant depression. We consider Spravato for these patients when traditional approaches haven’t been enough.
The Experience
What Does Spravato Treatment Feel Like?
Here’s what to actually expect during and after a Spravato session at our Rockville office.
During the Spray
5 Minutes
Our staff administers the nasal spray — two or three devices depending on your dose. It has a mild bitter taste. Some people feel a slight burning in the nose. The whole process takes about 5 minutes.
Monitoring Period
2 Hours
You stay in our office while the medication takes effect. Common feelings: mild dissociation, light-headedness, drowsiness, or a sense of “floating.” Some patients describe it as a dreamlike state. It typically peaks around 40 minutes and fades by the 2-hour mark.
After Treatment
Rest of the Day
Someone drives you home. Don’t drive, operate machinery, or make important decisions until the next morning. Most patients feel normal by the next day. Some notice a genuine improvement in mood surprisingly quickly.
Spravato Treatment Schedule
Weeks 1–4 (Induction): Two sessions per week at our office. This is when the medication builds its initial therapeutic effect.
Weeks 5–8 (Transition): Once per week. Your provider assesses response and adjusts dosing.
Week 9+ (Maintenance): Once every week or every two weeks, based on how you’re doing. Some patients eventually taper to once per month.
Comparisons
Spravato vs Ketamine: What’s the Difference?
This is the most common question we hear. Spravato and ketamine are related — but they’re not the same treatment. Here’s how they compare.
|
Spravato (Esketamine) |
IV Ketamine Infusion |
TMS Therapy |
|
|---|---|---|---|
|
What It Is |
Nasal spray — FDA-approved esketamine |
IV drip of racemic ketamine |
Magnetic pulses to the brain |
|
FDA Status |
FDA-approved for TRD & suicidal ideation |
Not FDA-approved for depression (off-label use) |
FDA-approved for depression & OCD |
|
Administration |
Clinician-administered nasal spray, in-office |
IV infusion in a clinic, 40–60 min |
Device placed on head, 20–30 min |
|
Insurance |
Covered by most major plans, Medicaid & Medicare |
Rarely covered — typically out-of-pocket |
Covered by most major plans |
|
Speed of Effect |
Hours to days |
Hours to days |
2–3 weeks typically |
|
Schedule |
2x/week → 1x/week → maintenance |
6 infusions over 2–3 weeks + boosters |
5 days/week for 4–6 weeks |
|
Monitoring |
2-hour in-office observation required |
Observation required during infusion |
None — drive yourself home |
|
Typical Cost |
Copay only with insurance |
$400–$800 per infusion (self-pay) |
Copay only with insurance |
The short version: Spravato is the only FDA-approved form of ketamine-based treatment for depression.
It’s a nasal spray rather than an IV, and it’s covered by insurance.
That combination — clinical approval, nasal delivery, and insurance coverage — is why it’s become the standard at practices like ours.
We accept major insurance providers
including Medicare & Medicaid








Don’t see your plan?
Call us. We may still be able to help.
Safety
Spravato Side Effects
Most side effects are temporary and happen during or shortly after the 2-hour monitoring period. Here’s the honest breakdown.
Common Side Effects
Pricing & Insurance
How Much Does Spravato Cost?
Insurance coverage is the biggest factor. Here’s what to expect.
Spravato Cost with Insurance
$0 – $35
per session (with copay card + insurance)
Most major insurance plans cover Spravato once treatment-resistant depression is documented. The manufacturer also offers a Spravato savings card that can reduce your copay to as low as $10 per session. We handle prior authorization for you.
Spravato Cost Without Insurance
$590 – $890
per session (varies by dose — 56mg vs 84mg)
Without insurance, Spravato is expensive. This is one of its key advantages over IV ketamine, though — because insurance actually covers it. Not sure about your coverage? Call us and we’ll check before your first visit.
Does Medicaid or Medicare Cover Spravato?
Yes — both Medicaid and Medicare cover Spravato in most cases. Medicare Part B covers it as an in-office administered medication. Medicaid coverage varies by state but is available in Maryland. We verify coverage and handle all paperwork on your behalf.

Meet Your Doctor
Dr. Amir Etesam, MD
Psychiatrist & Medical DirectorDr. Amir Etesam, MD, is a Johns Hopkins–trained, Board-Certified, psychiatrist and the founder of Bright Horizons Psychiatry in North Bethesda, Maryland. He is a distinguished member of Alpha Omega Alpha (AOA), the nation’s most respected medical honor society, recognizing excellence in scholarship and integrity in medicine.
Dr. Etesam’s clinical work is exclusively focused on patients with treatment-resistant major depressive disorder (TRD)—those who have not improved despite multiple trials of medication and therapy. His practice centers on advanced neuromodulation therapies, including Transcranial Magnetic Stimulation (TMS) and Intranasal Esketamine (Spravato®), provided in a private, state-of-the-art setting designed for comfort and healing.
He no longer accepts new patients for other psychiatric conditions such as anxiety, ADHD, bipolar disorder, or OCD.
For patients seeking care for other psychiatric conditions—such as anxiety, ADHD, bipolar disorder, and general mood or behavioral concerns—a highly trained team of psychiatric nurse practitioners works side-by-side with, and under the close supervision of Dr. Etesam. This collaborative model ensures that every patient receives the same standard of thoughtful, evidence-based care that defines Bright Horizons Psychiatry.
Common Questions
Frequently Asked Questions
About Spravato
Ready to Get Started?
Find out if Spravato
is right for you.
Free 30-minute consultation. We’ll tell you honestly whether Spravato makes sense for your situation — or if another treatment would be a better fit.


